CasNo:106-23-0
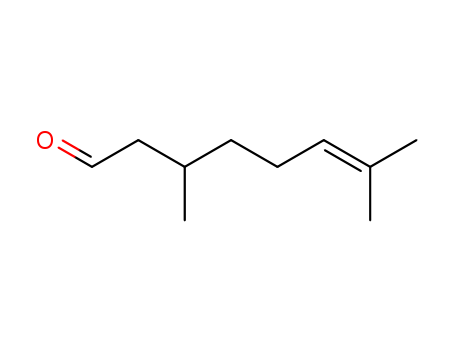
Product Name:Citronellal
Molecular Formula:C10H18O
Appearance:clear light yellow liquid
Purity:99%
Hot Sale! Chinese Factory Supply Citronellal 106-23-0 with Safe Delivery
- Molecular Formula:C10H18O
- Molecular Weight:154.252
- Appearance/Colour:clear light yellow liquid
- Vapor Pressure:0.215mmHg at 25°C
- Melting Point:-16°C (estimate)
- Refractive Index:n20/D 1.451(lit.)
- Boiling Point:208.4 °C at 760 mmHg
- Flash Point:75.6 °C
- PSA:17.07000
- Density:0.85 g/cm3
- LogP:2.95790
Citronellal (Cas 106-23-0) Usage
|
Chemical Properties |
clear light yellow liquid |
|
Occurrence |
Citronellal is a major constituent of citronella oil, which is derived from various species of plants, including Cymbopogon nardus and Cymbopogon winterianus. Citronella oil is well-known for its insect-repellent properties and is commonly used in products like candles, lotions, and sprays to deter mosquitoes and other insects. |
|
Description |
Citronellal is a natural compound found in citronella oil and various essential oils, known for its lemony scent and insect-repellent properties. It has diverse applications, including its use as a fragrance ingredient and its potential in antifungal activities. |
|
Definition |
ChEBI: A monoterpenoid, the main component of citronella oil which gives it its distinctive lemon aroma. |
| Uses |
Citronellal is valued for its pleasant lemony scent and is often used as a fragrance ingredient in perfumes, soaps, and other cosmetic products. (+)-Citronellal is extracted from citronella oils via fractional distillation. When needed, it can be further purified using addition compounds, such as the bisulfite derivative. Citronellal is widely used in the fragrance and perfumery industry due to its fresh, lemony aroma. In addition to its fragrance uses, citronellal is sometimes employed as a flavoring agent in the food industry. |
|
Aroma threshold values |
Detection: 31 to 100 ppb |
|
Taste threshold values |
Taste characteristics at 10 ppm: floral, green, rosy and citrus-lemon. |
|
Flammability and Explosibility |
Nonflammable |
|
Synthesis |
Can be prepared by chemical synthesis or by fractional distillation of natural oils, such as citronella. Industrially prepared by hydrogenation of β-citronellol or by catalytic hydrogenation of citral; also in the laboratory by dehydration of hydroxydihydrocitronellal. |
InChI:InChI=1/C10H18O/c1-9(2)5-4-6-10(3)7-8-11/h5,8,10H,4,6-7H2,1-3H3/t10-/m1/s1
106-23-0 Relevant articles
Synthesis of a Dialuminum-Substituted Silicotungstate and the Diastereoselective Cyclization of Citronellal Derivatives
Yuji Kikukawa, Syuhei Yamaguchi, Yoshinao Nakagawa, Kazuhiro Uehara, Sayaka Uchida, Kazuya Yamaguchi, and Noritaka Mizuno*
, J. Am. Chem. Soc. 2008, 130, 47, 15872–15878
Compound 1 showed high catalytic activity for the intramolecular cyclization of citronellal derivatives such as (+)-citronellal (3) and 3-methylcitronellal (4) without formation of byproduct resulting from etherification and dehydration.
Preparation of well-defined dendrimer encapsulated ruthenium nanoparticles and their application as catalyst and enhancement of activity when utilised as SCILL catalysts in the hydrogenation of citral
Antonels, Nathan Charles,Meijboom, Reinout
, p. 148 - 152 (2014)
Silica supported dendrimer encapsulated ...
Further analysis of the antimicrobial activity of α-phenylseleno citronellal and α-phenylseleno citronellol
Francine N. Victoria b, Cátia S. Radatz a, Maraísa Sachini a, Raquel G. Jacob a, Diego Alves a, Lucielli Savegnago d, Gelson Perin a, Amanda S. Motta c, Wladimir P. Silva b, Eder J. Lenardão a
, Food Control Volume 23, Issue 1, January 2012, Pages 95-99
The major constituents of the essential oil were identified as being (R)-citronellal 2 (45.4%), citronellol 5 (7.7%), geraniol 6 (19%) and limonene 7 (5.2%). The citronellal used in this work was isolated from the essential oil by distillation under reduced pressure (4 kPa) and its purity confirmed by GC.
106-23-0 Process route
-
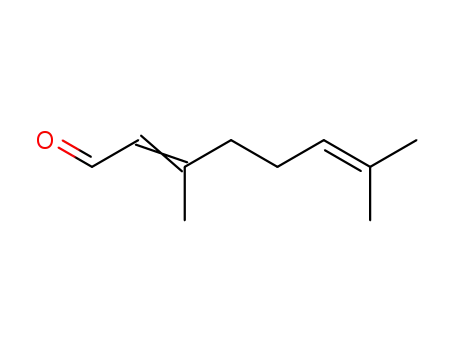
- 5392-40-5,96680-15-8
(E/Z)-3,7-dimethyl-2,6-octadienal

-
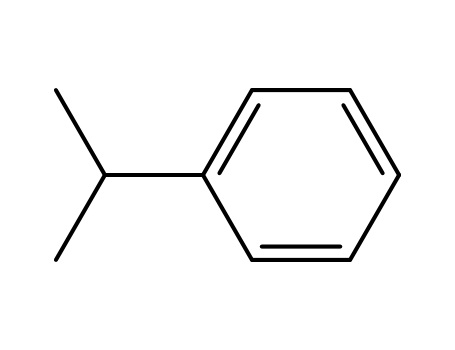
- 98-82-8
Isopropylbenzene

-
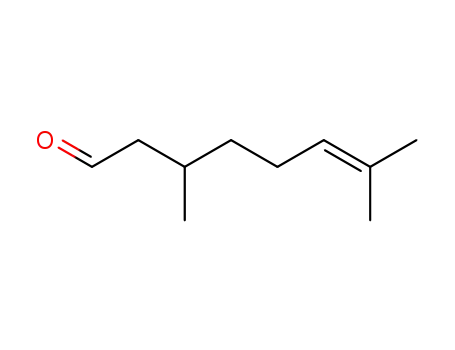
- 106-23-0,26489-02-1
3,7-dimethyl-oct-6-enal

-
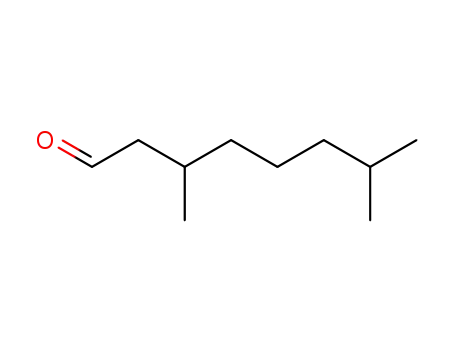
- 60018-13-5,81738-03-6,124440-53-5,25795-46-4,5988-91-0
3,7-dimethyl-1-octanal

-
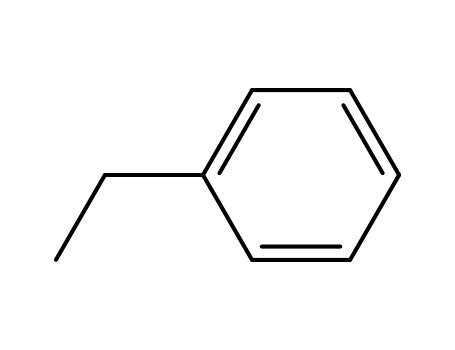
- 100-41-4,27536-89-6
ethylbenzene

-
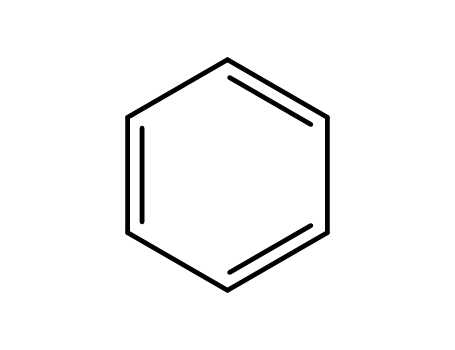
- 71-43-2,26181-88-4,54682-86-9,13967-78-7,174973-66-1
benzene
| Conditions | Yield |
|---|---|
|
With formic acid; palladium 10% on activated carbon; sodium formate; In water; at 80 ℃; for 0.5h; Reagent/catalyst; Solvent; Sealed tube; Microwave irradiation;
|
-
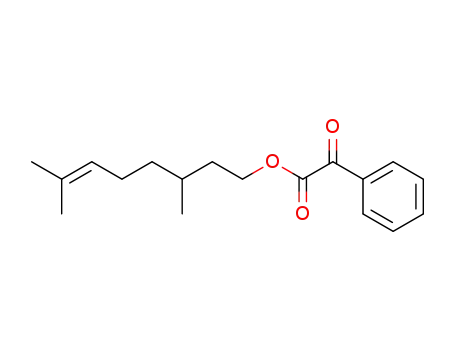
- 191750-82-0
(+/-)-3,7-dimethyloct-6-enyl 2-oxo-2-phenylacetate

-

- 106-23-0,26489-02-1
3,7-dimethyl-oct-6-enal

-
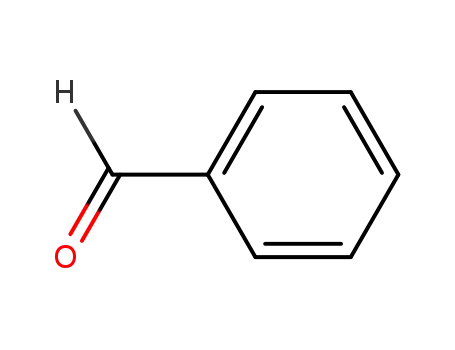
- 100-52-7
benzaldehyde

-
![(1S,9R)-6,10,10-Trimethyl-1-phenyl-3,11-dioxa-bicyclo[7.2.0]undecan-2-one](/upload/2023/8/622c78ac-b724-4006-87d9-3687da9c66c8.png)
-
(1S,9R)-6,10,10-Trimethyl-1-phenyl-3,11-dioxa-bicyclo[7.2.0]undecan-2-one
| Conditions | Yield |
|---|---|
|
In benzene; Irradiation;
|
70% |