CasNo:7331-52-4
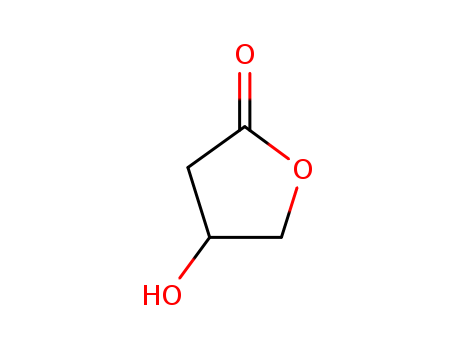
Product Name:(S)-3-Hydroxy-gamma-butyrolactone
Molecular Formula:C4H6O3
Appearance:Colorless to light yellow liquid
Purity:99%
Buy Reliable Quality Top Purity (S)-3-Hydroxy-gamma-butyrolactone 7331-52-4 with Safe Transportation
- Molecular Formula:C4H6O3
- Molecular Weight:102.09
- Appearance/Colour:Colorless to light yellow liquid
- Vapor Pressure:5.42E-05mmHg at 25°C
- Melting Point:1.24ºC
- Refractive Index:n20/D 1.464(lit.)
- Boiling Point:310.3 °C at 760 mmHg
- PKA:12.87±0.20(Predicted)
- Flash Point:157 °C
- PSA:46.53000
- Density:1.393 g/cm3
- LogP:-0.70580
(S)-3-Hydroxy-gamma-butyrolactone(Cas 7331-52-4) Usage
|
Description |
(S)-3-Hydroxy-gamma-butyrolactone is a chemical compound that plays a significant role in organic synthesis and serves as an important chiral pool. It finds application in the synthesis of various natural products, bioactive chiral drugs, and antibiotic chiral drugs. |
|
Chemical Properties |
Colorless to light yellow liquid. |
|
Uses |
(S)-3-Hydroxy-gamma-butyrolactone is an optically pure and essential chiral building block utilized in the pharmaceutical industry for synthesizing various drugs and compounds. (S)-3-Hydroxy-gamma-butyrolactone demonstrates anticancer drug resistance inhibition properties. It is also utilized as an organic solvent in the production of malic acid. Its synthesis typically involves the hydrolysis of butyrolactone, using aqueous hydrochloric acid and sodium chloride as an acidic catalyst. It can be deoxidized to produce (S)-(+)-3-Hydroxytetrahydrofuran, which acts as a crucial intermediate for anti-AIDS drugs. |
|
Precautions |
For best results, Store in cool, dry place in tightly closed containers, under inert gas and protected from moisture as this substance is moisture sensitive. (S)-3-Hydroxy-gamma-butyrolactone is incompatible with oxidizing agents. This chemical causes skin irritation and serious eye irritation. |
InChI:InChI=1/C4H6O3/c5-3-1-4(6)7-2-3/h3,5H,1-2H2/t3-/m0/s1
7331-52-4 Relevant articles
Ru/SiO2 Catalyst for Highly Selective Hydrogenation of Dimethyl Malate to 1,2,4-Butanetriol at Low Temperatures in Aqueous Solvent
Chen, Can,Jiang, Junxiang,Li, Guangci,Li, Xuebing,Wang, Da,Wang, Zhong,Yu, Pei
, (2022/01/12)
Catalytic selective hydrogenation of est...
Direct Conversion of (S)-3-Hydroxy-gamma-butyrolactone to Chiral Three-Carbon Building Blocks.
Wang G 1 , Hollingsworth RI
, The Journal of Organic Chemistry, 01 Feb 1999, 64(3):1036-1038
While optically active 3-hydroxy-gamma-butyrolactone(HGBL) is a useful C4 chiral synthon for the synthesis of chiral pharmaceuticals....
7331-52-4 Process route
-
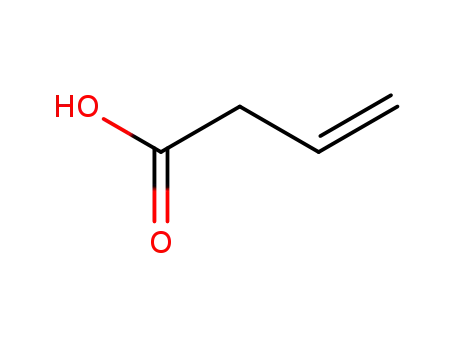
- 625-38-7
but-3-enoic acid

-
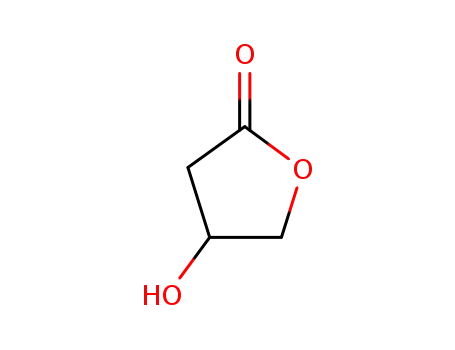
- 5469-16-9,7331-52-4,58081-05-3,131432-37-6
4-Hydroxy-dihydro-furan-2-on
| Conditions | Yield |
|---|---|
|
With dihydrogen peroxide; In tert-butyl alcohol;
|
91% |
|
With osmium(VIII) oxide; barium chlorate; water;
|
|
|
With Perbenzoic acid; chloroform;
|
-
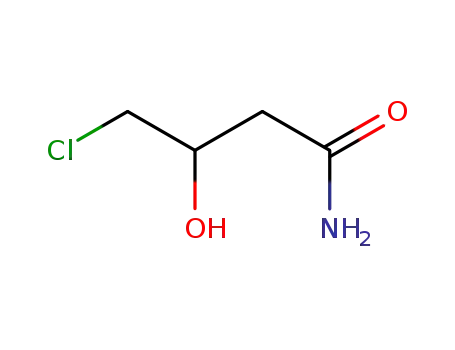
- 51499-71-9
4-chloro-3-hydroxybutylamide

-

- 5469-16-9,7331-52-4,58081-05-3,131432-37-6
4-Hydroxy-dihydro-furan-2-on
| Conditions | Yield |
|---|---|
|
In water; butanone; at 60 ℃; for 24h;
|
92% |
|
In water; at 70 ℃; for 3h;
|
65.2% |
|
In water; at 30 - 100 ℃; for 0.5 - 336h; pH=1.2 - 5.5; Aqueous phophate buffer;
|
56.5% |