CasNo:1948-33-0
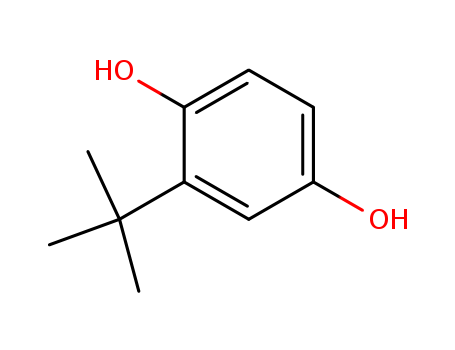
Product Name:tert-Butylhydroquinone
Molecular Formula:C10H14O2
Appearance:tan powder
Purity:99%
Factory Supply tert-Butylhydroquinone, Offer Quality 1948-33-0 with Reasonable Price
- Molecular Formula:C10H14O2
- Molecular Weight:166.22
- Appearance/Colour:tan powder
- Vapor Pressure:0.001mmHg at 25°C
- Melting Point:127-129 °C(lit.)
- Refractive Index:1.559
- Boiling Point:291.3 °C at 760 mmHg
- PKA:10.80±0.18(Predicted)
- Flash Point:138.7 °C
- PSA:40.46000
- Density:1.086 g/cm3
- LogP:2.39530
tert-Butylhydroquinone(Cas 1948-33-0) Usage
| General Description |
tert-Butylhydroquinone (TBHQ) is a white, crystalline solid with a characteristic odor. It is a versatile antioxidant used in the preservation of fats, oils, and food products, with additional applications in research and materials science. TBHQ can be found naturally in vegetable oils, animal fats, varnishes, lacquers, resins, oil field additives, and perfumes. |
|
Uses |
tert-Butylhydroquinone (TBHQ) exhibits excellent stabilizing effects in unsaturated fats and oils. It serves as an antioxidant and is commonly used to preserve oils, fats, and various food items. TBHQ has also been utilized in the development of environmentally friendly electrode materials for supercapacitors. By decorating the surface of graphene nanosheets with TBHQ, enhanced properties for supercapacitors have been achieved. |
|
Definition |
ChEBI: A member of the class of hydroquinones in which one of the ring hydrogens of hydroquinone is replaced by a tert-butyl group. |
|
Air & Water Reactions |
Insoluble in water. |
|
Fire Hazard |
tert-Butylhydroquinone is combustible. |
|
Flammability and Explosibility |
Nonflammable |
|
Purification Methods |
Recrystallise the hydroquinone from H2O or MeOH and dry it in a vacuum at 70o. Store it in a dark container. [Stroh et al. Angew Chem 69 699 1957, Beilstein 6 IV 6013.] |
|
Who Evaluation |
Evaluation year: 1997 |
InChI:InChI=1/C10H12O3/c1-10(2,3)8-6(11)4-5-7(12)9(8)13/h4-5,13H,1-3H3
1948-33-0 Relevant articles
Chemoprotective and Carcinogenic Effects of tert-Butylhydroquinone and Its Metabolites
Gharavi, Negar; Haggarty, Susan; S. El-Kadi, Ayman O.
, Current Drug Metabolism, Volume 8, Number 1, 2007, pp. 1-7(7)
Tert-butylhydroquinone (tBHQ) has been commonly used as a synthetic food antioxidant to prevent oils and fats from oxidative deterioration and rancidity due to its potent anti-lipid peroxidation activity. In North America, the maximum level of tBHQ allowed in fat products is 0.02% with an acceptable daily intake of 0 - 0.7 mg/kg body weight. Extensive studies have demonstrated that tBHQ exhibit anti-carcinogenic effect.
Sulfonic acid functionalized MCM-41 as solid acid catalyst for tert-butylation of hydroquinone enhanced by microwave heating
Ng, Eng-Poh,Mohd Subari, Siti Norbayu,Marie, Olivier,Mukti, Rino R.,Juan, Joon-Ching
, p. 34 - 41 (2013)
Covalently linked sulfonic acid (SO3H) m...
Cell death induced by the phenolic antioxidant tert-butylhydroquinone and its metabolite tert-butylquinone in human monocytic leukemia U937 cells
T Okubo a 1, Y Yokoyama b, K Kano b, I Kano a 2
, Food and Chemical Toxicology Volume 41, Issue 5, May 2003, Pages 679-688
2-tert-Butylhydroquinone (TBHQ) is a strong antioxidant that is used as a food additive in oils, fat and meat products, primarily to prevent rancidity. Metabolically, TBHQ is formed from 3-tert-butyl-4-hydroxyanisole (BHA), another widely used food additive, by O-demethylation (Armstrong and Wattenberg, 1985), and it is further oxidized to 2-tert-butyl-1,4-benzoquinone (TBQ) (Kahl et al., 1989).
1948-33-0 Process route
-
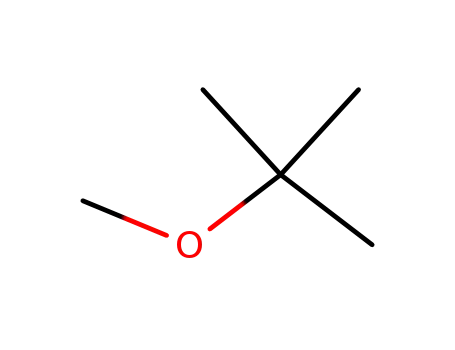
- 1634-04-4
tert-butyl methyl ether

-
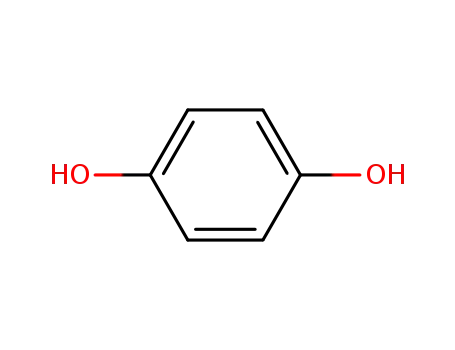
- 123-31-9,8027-02-9
hydroquinone

-
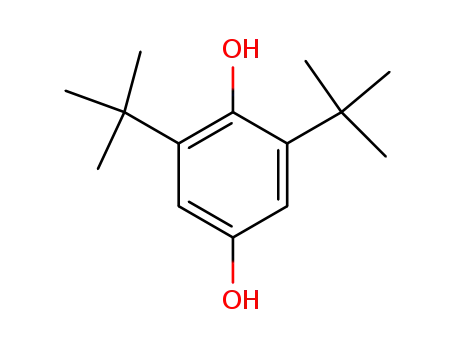
- 2444-28-2
2,6-di-tert-butyl-4-hydroxyphenol

-
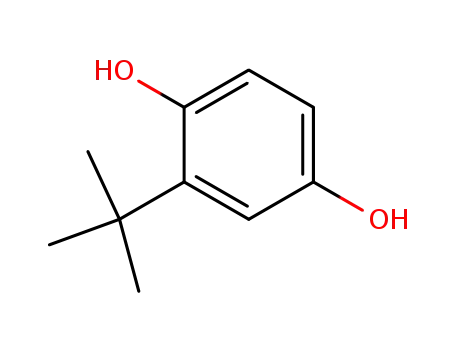
- 1948-33-0
tert-butylhydroquinone
| Conditions | Yield |
|---|---|
|
With 3-(4-sulfobutylamino)propylsilanized MCM-41; In nitrobenzene; at 165 ℃; for 0.166667h; chemoselective reaction; Microwave irradiation; Green chemistry;
|
-

- 1634-04-4
tert-butyl methyl ether

-

- 123-31-9,8027-02-9
hydroquinone

-

- 2444-28-2
2,6-di-tert-butyl-4-hydroxyphenol

-
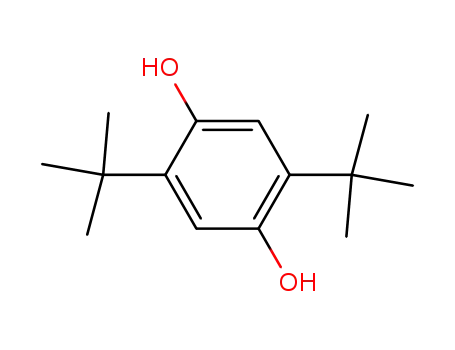
- 88-58-4
2,5-bis(1,1-dimethylethyl)-1,4-benzenediol

-
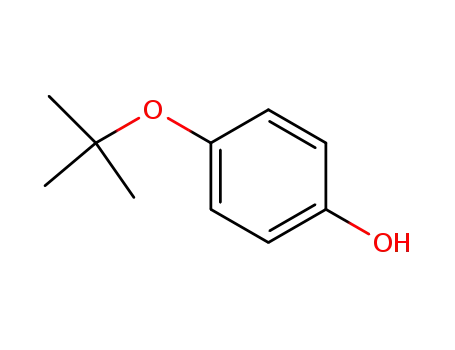
- 2460-87-9
4-(tert-butoxy)phenol

-

- 1948-33-0
tert-butylhydroquinone
| Conditions | Yield |
|---|---|
|
With 3-(4-sulfobutylamino)propylsilanized MCM-41; In nitrobenzene; at 150 ℃; for 0.133333h; Concentration; chemoselective reaction; Catalytic behavior; Microwave irradiation; Green chemistry;
|