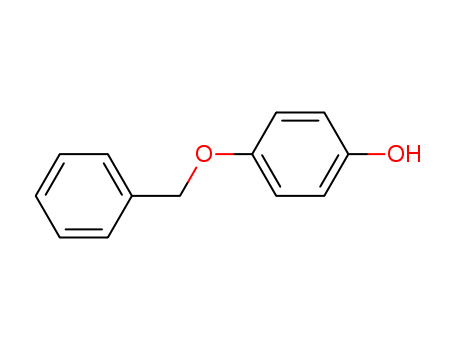
CasNo:103-16-2
Product Name:Monobenzone
Molecular Formula:C13H12O2
Appearance:cream to beige-light brownish crystals
Purity:99%
Buy Reliable Quality Monobenzone 103-16-2, Best Quality with Safe Shipping
- Molecular Formula:C13H12O2
- Molecular Weight:200.237
- Appearance/Colour:cream to beige-light brownish crystals
- Melting Point:119-120 °C(lit.)
- Refractive Index:1.5906 (estimate)
- Boiling Point:359.1 °C at 760 mmHg
- PKA:10.29±0.15(Predicted)
- Flash Point:213.4 °C
- PSA:29.46000
- Density:1.16 g/cm3
- LogP:2.97120
Monobenzone (Cas 103-16-2) Usage
|
Description |
Monobenzone, also known as 4-(Benzyloxy)phenol and monobenzyl ether of hydroquinone (MBEH), is an organic chemical compound in the phenol family with the chemical formula C6H5CH2OC6H4OH. When applied topically, Monobenzone increases the excretion of melanin from melanocytes in the skin. This action is thought to be responsible for its depigmenting effect in humans. |
|
Uses |
Monobenzone is classified as a hydroquinone monobenzyl ether. Monobenzone was initially introduced for the treatment of disorders involving excess melanin pigmentation, such as melasma. However, it is now primarily used to permanently depigment normally pigmented skin in patients with extensive vitiligo, a skin condition characterized by the loss of skin pigmentation. |
|
Brand name |
Benoquin (Valeant). |
InChI:InChI=1/C14H14O.C6H6O2/c1-3-7-13(8-4-1)11-15-12-14-9-5-2-6-10-14;7-5-1-2-6(8)4-3-5/h1-10H,11-12H2;1-4,7-8H
103-16-2 Relevant articles
Skin-Depigmenting Agent Monobenzone Induces Potent T-Cell Autoimmunity toward Pigmented Cells by Tyrosinase Haptenation and Melanosome Autophagy
Jasper G. van den Boorn 1, Daisy I. Picavet 1, Paul F. van Swieten 2, Henk A. van Veen 3, Debby Konijnenberg 1, Peter A. van Veelen 4, Toni van Capel 3, Esther C de Jong 3, Eric A. Reits 3, Jan W. Drijfhout 4, Jan D. Bos 1, Cornelis J.M. Melief 4, Rosalie M. Luiten 1
, Journal of Investigative Dermatology Volume 131, Issue 6, June 2011, Pages 1240-1251
Monobenzone further augments the processing and shedding of melanocyte-differentiation antigens by inducing melanosome autophagy and enhanced tyrosinase ubiquitination, ultimately activating dendritic cells, which induced cytotoxic human melanoma-reactive T cells.
Dermatitis Produced by Applications of Monobenzone in Patients With Active Vitiligo
James J. Nordlund, MD; Bernadette Forget, RN, MSN; John Kirkwood, MD; Aaron B. Lerner, MD
, Arch Dermatol. 1985;121(9):1141-1144
We have developed a catalytic electron t...
103-16-2 Process route
-
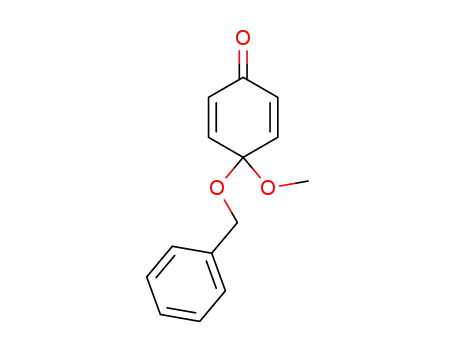
- 73010-55-6
4-(benzyloxy)-4-methoxy-2,5-cyclohexadien-1-one

-
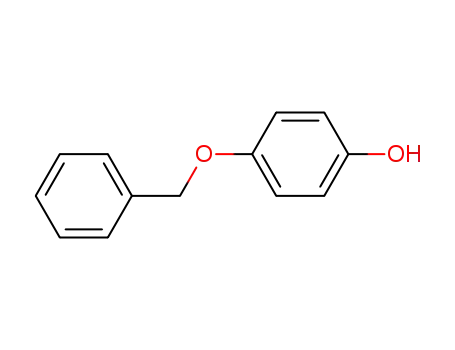
- 103-16-2
4-Benzyloxyphenol

-
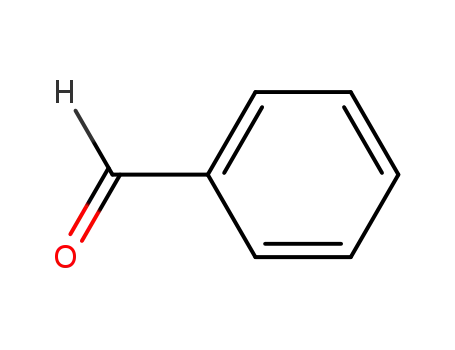
- 100-52-7
benzaldehyde

-
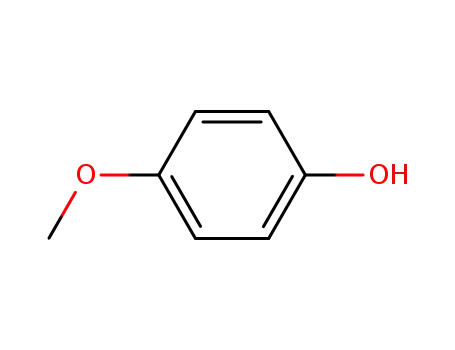
- 150-76-5
4-methoxy-phenol
| Conditions | Yield |
|---|---|
|
In tetrahydrofuran; at 180 ℃; for 4.5h;
|
-
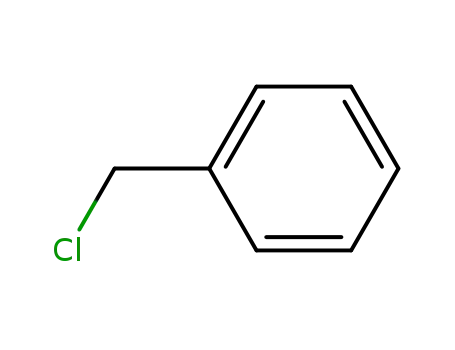
- 100-44-7
benzyl chloride

-
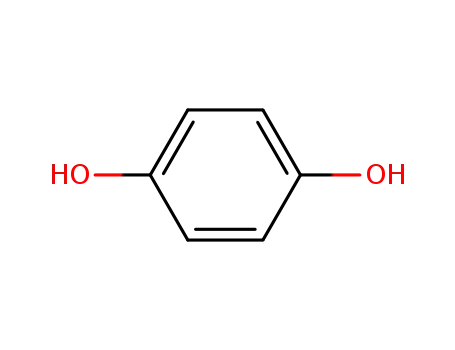
- 123-31-9,8027-02-9
hydroquinone

-

- 103-16-2
4-Benzyloxyphenol

-
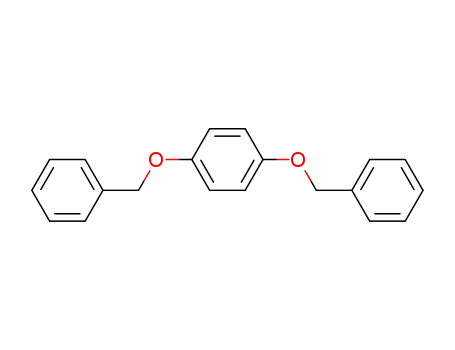
- 621-91-0
1,4-dibenzyloxybenzene
| Conditions | Yield |
|---|---|
|
With xylene;
|
|
|
With potassium carbonate; acetone;
|
|
|
With ethanol; sodium ethanolate;
|
|
|
With ethanol; anion-exchanger;
|
103-16-2 Upstream products
-
100-39-0
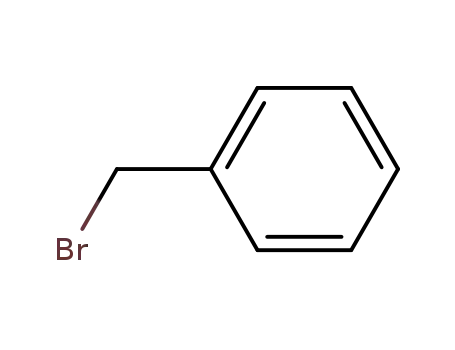
benzyl bromide
-
123-31-9

hydroquinone
-
100-44-7
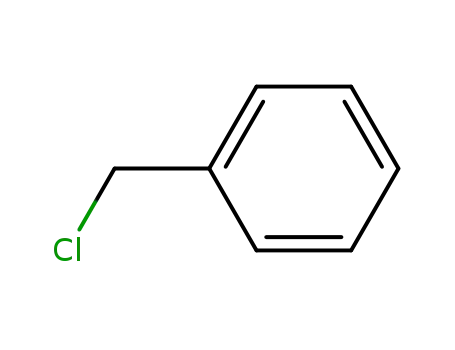
benzyl chloride
-
6630-18-8
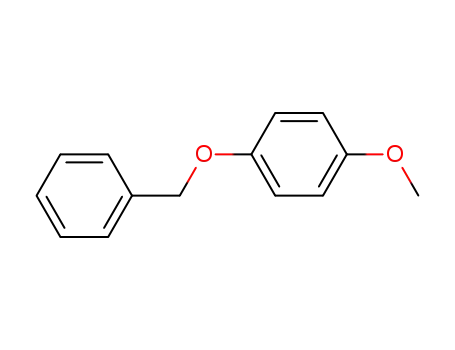
4-methoxyphenyl benzyl ether
103-16-2 Downstream products
-
119-98-2
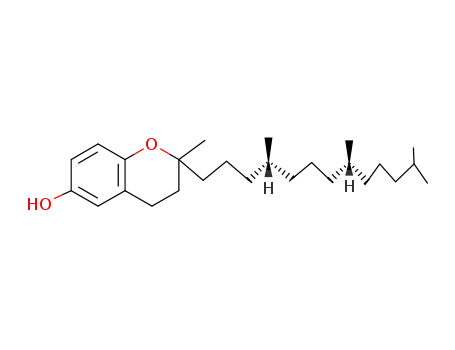
(2RS,4'R,8'R)-Tocol
-
625851-45-8
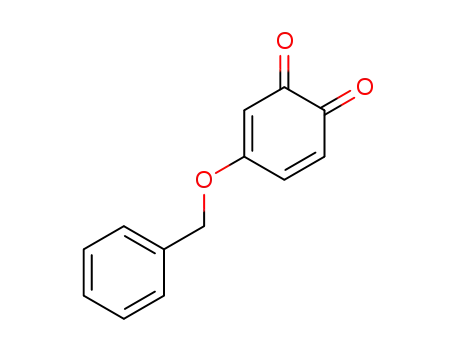
4-benzyloxy-[1,2]benzoquinone
-
102079-74-3
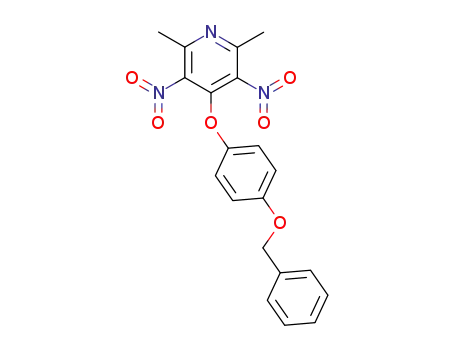
4-(4-benzyloxy-phenoxy)-2,6-dimethyl-3,5-dinitro-pyridine
-
101936-55-4
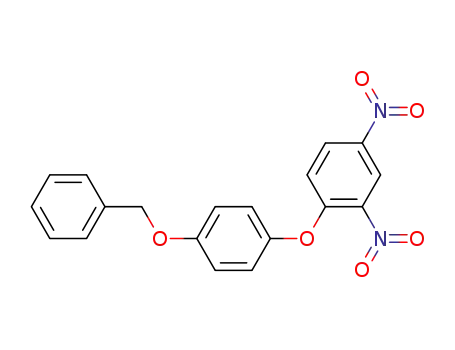
1-benzyloxy-4-(2,4-dinitro-phenoxy)-benzene