CasNo:125-71-3
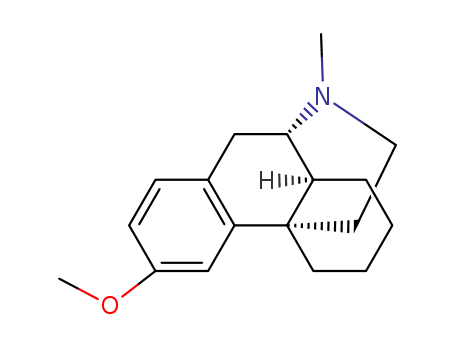
Product Name:Dextromethorphan
Molecular Formula:C18H25NO
Purity:99%
Wholesale Factory Supply Dextromethorphan 125-71-3 with Efficient Transportation
- Molecular Formula:C18H25NO
- Molecular Weight:271.403
- Vapor Pressure:1.92E-06mmHg at 25°C
- Melting Point:80 °C
- Refractive Index:1.585
- Boiling Point:394.9 °C at 760 mmHg
- PKA:pKa 8.3 (Uncertain)
- Flash Point:116.2 °C
- PSA:12.47000
- Density:1.11 g/cm3
- LogP:3.32130
DEXTROMETHORPHAN(Cas 125-71-3) Usage
|
Description |
Dextromethorphan is structurally related to opioids but is devoid of analgesic or sedative effects at therapeutic doses. It is metabolized in the liver, primarily by the enzyme CYP2D6, into a more potent metabolite called dextrorphan. |
|
Uses |
Dextromethorphan is used as a non-prescription cough suppressant and antitussive. It is employed to treat non-productive cough (coughing without mucus production). Dextromethorphan's metabolite, dextrorphan, is a potent noncompetitive antagonist of the N-methyl-D-aspartate (NMDA) glutamate receptor. Dextromethorphan's NMDA receptor antagonism has led to its use in treating phantom pain. |
|
Brand name |
Benylin DM (Parke-Davis); Dextromethorphan Hydrobromide OROS Tablets (Ciba-Geigy); Drixoral Cough (Schering-Plough HealthCare); PediaCare 1 (McNeil Consumer); Romilar (Hoffmann-LaRoche-International); St. Joseph Cough Syrup (Schering-Plough HealthCare);Agrippol;Dextophan;Dextrophen. |
|
Biological Functions |
Dextromethorphan acts on the central nervous system to suppress the cough reflex. |
InChI:InChI=1/C18H25NO/c1-19-10-9-18-8-4-3-5-15(18)17(19)11-13-6-7-14(20-2)12-16(13)18/h6-7,12,15,17H,3-5,8-11H2,1-2H3/t15-,17+,18+/m1/s1
125-71-3 Relevant articles
Synthesis, in vitro and in vivo studies, and molecular modeling of N-alkylated dextromethorphan derivatives as non-competitive inhibitors of α3β4 nicotinic acetylcholine receptor
Jozwiak, Krzysztof,Targowska-Duda, Katarzyna M.,Kaczor, Agnieszka A.,Kozak, Joanna,Ligeza, Agnieszka,Szacon, Elzbieta,Wrobel, Tomasz M.,Budzynska, Barbara,Biala, Grazyna,Fornal, Emilia,Poso, Antti,Wainer, Irving W.,Matosiuk, Dariusz
, p. 6846 - 6856 (2014)
9 N-alkylated derivatives of dextrometho...
Dextromethorphan decreases the excitability of the human motor cortex
Ulf Ziemann, Robert Chen, Leonardo G. Cohen, Mark Hallett
, neurology, November 01, 1998; 51 (5)
Dextromethorphan (DM), a noncompetitive N-methyl-D-aspartate receptor antagonist, has recently attracted clinical interest for its potential as a neuroprotective agent in various models of excitotoxicity. We were interested in learning whether this drug can modulate the excitability of the motor cortex in healthy subjects.
Contra-thermodynamic Hydrogen Atom Abstraction in the Selective C-H Functionalization of Trialkylamine N-CH3 Groups
Barham, Joshua P.,John, Matthew P.,Murphy, John A.
supporting information, p. 15482 - 15487 (2016/12/09)
We report a simple one-pot protocol that...
125-71-3 Process route
-

- 186581-53-3,908094-01-9,334-88-3
diazomethane

-
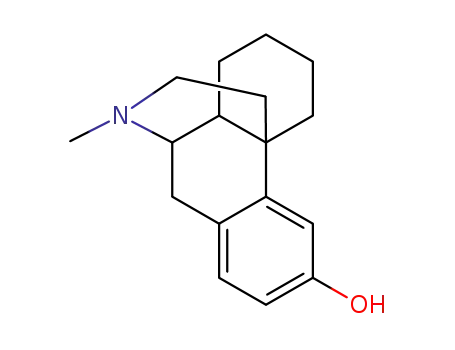
- 888939-18-2
3-Hydroxy-N-methylmorphinan

-
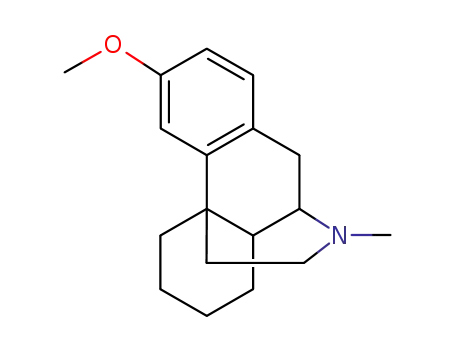
- 125-70-2,125-71-3,510-53-2,3561-92-0,1453167-99-1
dextromethorphan
| Conditions | Yield |
|---|---|
|
In methanol; chloroform; water; for 15h; Ambient temperature;
|
95% |
-
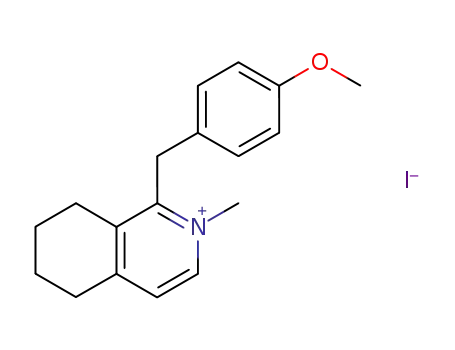
- 98237-33-3
1-(4-Methoxybenzyl)-2-methyl-5,6,7,8-tetrahydroisochinoliniumiodide

-

- 125-70-2,125-71-3,510-53-2,3561-92-0,1453167-99-1
dextromethorphan
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 3 steps
1: NaBH4, NaOH / aq. ethanol / 15 h / Ambient temperature
2: 79 percent / 48percent aq. HBr / 40 h / 140 °C
3: 95 percent / CHCl3; methanol; H2O / 15 h / Ambient temperature
With sodium hydroxide; sodium tetrahydroborate; hydrogen bromide; In methanol; ethanol; chloroform; water;
|