CasNo:147-24-0
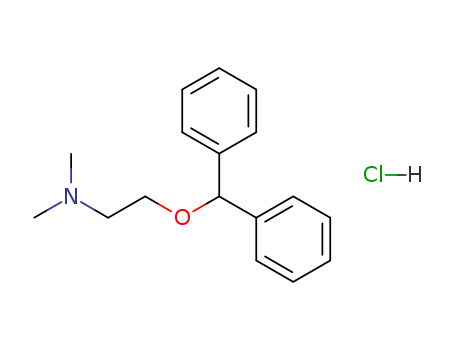
Product Name:Diphenhydramine Hydrochloride
Molecular Formula:C17H21NO.HCl
Appearance:White crystalline powder
Purity:99%
99% Pure Buy Quality Diphenhydramine Hydrochloride 147-24-0 Reasonable Price
- Molecular Formula:C17H21NO.HCl
- Molecular Weight:291.821
- Appearance/Colour:White crystalline powder
- Melting Point:168-172 °C
- Refractive Index:1.5800 (estimate)
- Boiling Point:343.7 °C at 760 mmHg
- Flash Point:101.5 °C
- PSA:12.47000
- Density:1.024 g/cm3
- LogP:4.15620
Diphenhydramine hydrochloride(Cas 147-24-0) Usage
|
Description |
Diphenhydramine hydrochloride is an antihistamine drug having the chemical name 2-(Diphenylmethoxy)- N,N-dimethylethylamine hydrochloride. It occurs as a white, crystalline powder, is freely soluble in water and alcohol and has a molecular weight of 291.82. The molecular formula is C17H21NO·HCl and the structural formula is as follows: Diphenhydramine hydrochloride in the parenteral form is a sterile, pyrogen-free solution available in a concentration of 50 mg of diphenhydramine hydrochloride per mL. The solutions for parenteral use have been adjusted to a pH between 5.0 and 6.0 with either sodium hydroxide or hydrochloric acid and contains 0.1 mg/mL benzethonium chloride as a germicidal agent. |
|
Clinical Pharmacology |
Diphenhydramine hydrochloride is an antihistamine with anticholinergic (drying) and sedative side effects. Antihistamines appear to compete with histamine for cell receptor sites on effector cells. Diphenhydramine hydrochloride in the injectable form has a rapid onset of action. Diphenhydramine hydrochloride is widely distributed throughout the body, including the CNS. A portion of the drug is excreted unchanged in the urine, while the rest is metabolized via the liver. Detailed information on the pharmacokinetics of Diphenhydramine Hydrochloride Injection is not available. |
|
Indications and Usage |
Diphenhydramine hydrochloride in the injectable form is effective in adults and pediatric patients, other than premature infants and neonates, for the following conditions when diphenhydramine hydrochloride in the oral form is impractical. antihistaminic: For amelioration of allergic reactions to blood or plasma, in anaphylaxis as an adjunct to epinephrine and other standard measures after the acute symptoms have been controlled, and for other uncomplicated allergic conditions of the immediate type when oral therapy is impossible or contraindicated. Motion Sickness: For active treatment of motion sickness. Antiparkinsonism: For use in parkinsonism, when oral therapy is impossible or contraindicated, as follows: parkinsonism in the elderly who are unable to tolerate more potent agents; mild cases of parkinsonism in other age groups, and in other cases of parkinsonism in combination with centrally acting anticholinergic agents. |
|
Drug overdose |
Antihistamine overdosage reactions may vary from central nervous system depression to stimulation. Stimulation is particularly likely in pediatric patients. Atropine-like signs and symptoms; dry mouth; fixed, dilated pupils; flushing; and gastrointestinal symptoms may also occur. Stimulants should not be used. Vasopressors may be used to treat hypotension. |
|
Contraindications |
Use in Neonates or Premature Infants: This drug should not be used in neonates or premature infants. Use in Nursing Mothers: Because of the higher risk of antihistamines for infants generally, and for neonates and prematures in particular, antihistamine therapy is contraindicated in nursing mothers. Use as a Local Anesthetic: Because of the risk of local necrosis, this drug should not be used as a local anesthetic. Antihistamines are also contraindicated in the following conditions: Hyper sensitivity to diphenhydramine hydrochloride and other antihistamines of similar chemical structure. |
|
Chemical Properties |
White or almost white, crystalline powder. odorless and has a bitter, numbing taste. soluble in water and in alcohol. |
|
Originator |
Benadryl,Parke Davis,US,1946 |
|
Uses |
Diphenhydramine is a H1-histamine receptor antagonist with anticholinergic (drying) and sedative side effects. It is categorized as an antihistaminic; sedative, hypnotic, treatment of allergic. It is used to treat allergy and cold symptoms such as sneezing, runny nose, watery eyes, urticaria (hives), skin rash, and pruritus (itchy skin). |
|
Preparation |
Diphenhydramine Hydrochloride synthesis is a four-step sequence starting with a Grignard reaction. Synthesis of Diphenhydramine Hydrochloride 6.A) modified synthetic route to diphenhydramine hydrochloride.B) Sequence of unit operations required for the synthesis. The dotted boxes denote the four stages of the synthesis. DMAE: dimethylaminoethanol.Organic synthesis in a modular robotic system driven by a chemical programming language |
|
Application |
Diphenhydramine hydrochloride is used as an antihistamine and for its antieholinergie (drying) and sedative effects; for allergic conjunctivitis due to foods; for mild, uncomplicated allergic skin manifestations of urticaria and angioedema; for amelioration of allergic reactions to blood or plasma; for dermatographism; in therapy for anaphylaetie reactions adjunctive to epinephrine and other standard measures after the acute manifestations have been controlled; for active and prophylactie treatment of motion sickness; for parkinsonism; an antiemetic; has local anesthetic properties; in preparations for the relief of cough; in the prevention and treatment of radiation sickness, nausea and vomiting; for treatment of allergie cheilitis and stomatitis. |
|
Definition |
ChEBI: Diphenhydramine Hydrochloride is the hydrochloride salt form of diphenhydramine, an ethanolamine and first-generation histamine antagonist with anti-allergic activity. |
|
Brand name |
Benadryl (Parke-Davis). |
|
Therapeutic Function |
Antihistaminic |
|
General Description |
Diphenhydramine hydrochloride(Benadryl), is an oily, lipid-soluble free base available as the bitter-tasting hydrochloride salt, which is a stable, White or almost-white crystalline powder. Odorless with a bitter numbing taste. soluble in water (1:1), alcohol (1:2) and chloroform (1:2). The salt has a pKa value of 9, and a 1% aqueous solution has a pH of about 5. |
|
Air & Water Reactions |
Water soluble. Aqueous solutions are acidic. |
|
Reactivity Profile |
N-(2-Diphenylmethoxyethyl)-N,N-dimethylamine hydrochloride gives acidic solutions in water. Neutralizes bases. May react with strong oxidizing and strong reducing agents. May catalyze organic reactions. Slowly darkens on exposure to light. |
|
Health Hazard |
Potentially. If taken in large quantities, diphenhydramine can cause severe agitation and confusion, fever, skin flushing, problems with vision, dry mouth, dry eyes, and inability to sweat. Overdoses can lead to high heart rates, abnormal heart rhythms, seizures, and death.If given to elderly patients, diphenhydramine can cause confusion and agitation. Because of this, diphenhydramine is not recommended in elderly patients for insomnia or treatment for the common cold; though it should still be given in cases of allergic reaction. |
|
Biological Activity |
Diphenhydramine (DPH) is a first generation antihistamine that is a potent antagonist of the histamine H1 receptor (Ki = 11.7 nM using human recombinant receptors). DPH readily crosses the blood-brain barrier and produces diverse cognitive and psychomotor effects. DPH also antagonizes muscarinic cholinergic receptors (Kis = 100 to 260 nM for M1-M5), increasing the range of central nervous system effects and applications. |
|
Contact allergens |
This antihistaminic drug with sedative properties is mainly sold over the counter. It can be used both topically (treatment of pruritis) and orally for its antiallergic, antiemetic, sedative, and anticough properties. Allergic or photoallergic contact dermatitis and fixeddrug eruption seem to be rare. |
|
Biochem/physiol Actions |
Diphenhydramine hydrochloride (DPH) is an antihistaminic agent that relieves symptoms of hypersensitive reactions. It exhibits antimuscarinic and marked sedative effects. DPH is also used to prevent nausea, vomiting and vertigo of various causes. In addition, it acts as a potential therapeutic for insomnia. |
|
Safety Profile |
Poison by ingestion, subcutaneous, intravenous, and intraperitoneal routes. Human systemic effects by ingestion or skin contact: arrhythmias, ataxia, blood pressure elevation, convulsions, distorted perceptions, eye effects, and hallucinations. Experimental teratogenic and reproductive effects. Questionable carcinogen with experimental tumorigenic data. When heated to decomposition it emits very toxic fumes of NO, and HCl. See also ESTERS and ETHERS. |
|
Veterinary Drugs and Treatments |
In veterinary medicine, diphenhydramine is used principally for its antihistaminic effects, but also for other pharmacologic actions. Its sedative effects can be of benefit in treating the agitation (pruritus, etc.) associated with allergic responses. It has also been used for treatment and prevention of motion sickness and as an antiemetic in small animals. It has been suggested for use as adjunctive treatment of aseptic laminitis in cattle and it may be useful as an adjunctive treatment for feline pancreatitis. For other suggested uses, refer to the Dosage section below. |
InChI:InChI=1/C17H21NO.2ClH/c1-18(2)13-14-19-17(15-9-5-3-6-10-15)16-11-7-4-8-12-16;;/h3-12,17H,13-14H2,1-2H3;2*1H
147-24-0 Relevant articles
On-demand continuous-flow production of pharmaceuticals in a compact, reconfigurable system
Adamo, Andrea,Beingessner, Rachel L.,Behnam, Mohsen,Chen, Jie,Jamison, Timothy F.,Jensen, Klavs F.,Monbaliu, Jean-Christophe M.,Myerson, Allan S.,Revalor, Eve M.,Snead, David R.,Stelzer, Torsten,Weeranoppanant, Nopphon,Wong, Shin Yee,Zhang, Ping
, p. 61 - 67 (2016)
Pharmaceutical manufacturing typically u...
End-to-end continuous flow synthesis and purification of diphenhydramine hydrochloride featuring atom economy, in-line separation, and flow of molten ammonium salts
Snead, David R.,Jamison, Timothy F.
, p. 2822 - 2827 (2013)
A continuous end-to-end synthesis and pu...
Organic synthesis in a modular robotic system driven by a chemical programming language
Steiner, Sebastian,Wolf, Jakob,Glatzel, Stefan,Andreou, Anna,Granda, Jaros?aw M.,Keenan, Graham,Hinkley, Trevor,Aragon-Camarasa, Gerardo,Kitson, Philip J.,Angelone, Davide,Cronin, Leroy
, (2018/12/14)
The synthesis of complex organic compoun...
Method for preparing toluene benzyl phenyl substituted compound
-
Paragraph 0028; 0029; 0030; 0031, (2018/03/26)
In order to overcome the shortcomings of...
147-24-0 Process route
-
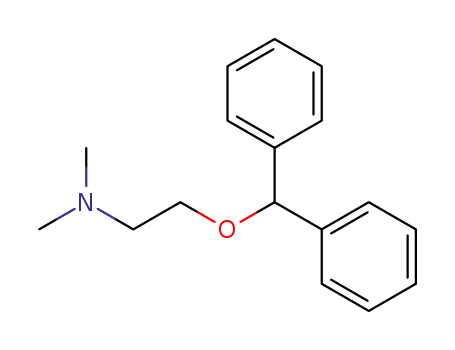
-
58-73-1
2-diphenylmethoxy-N,N-dimethylethanamine

-
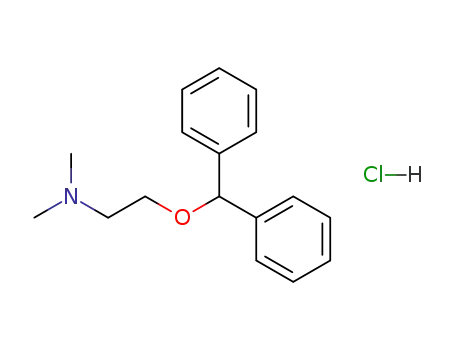
-
147-24-0,8052-21-9
diphenhydramine hydrochloride
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride;
In
ethanol;
at 20 ℃;
for 1h;
Time;
|
9.6 mmol |
-
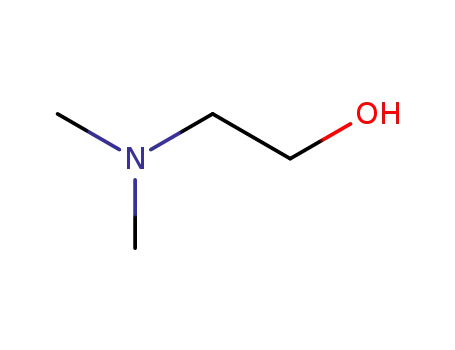
-
108-01-0
2-(N,N-dimethylamino)ethanol

-
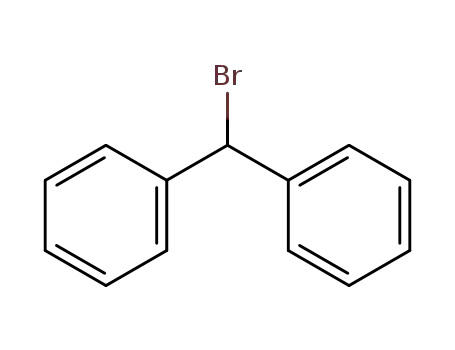
-
776-74-9
Bromodiphenylmethane

-

-
147-24-0,8052-21-9
diphenhydramine hydrochloride
| Conditions | Yield |
|---|---|
|
2-(N,N-dimethylamino)ethanol; Bromodiphenylmethane;
In
toluene;
for 20h;
Reflux;
With
hydrogenchloride;
In
diethyl ether;
at 25 ℃;
|
147-24-0 Upstream products
-
17616-20-5
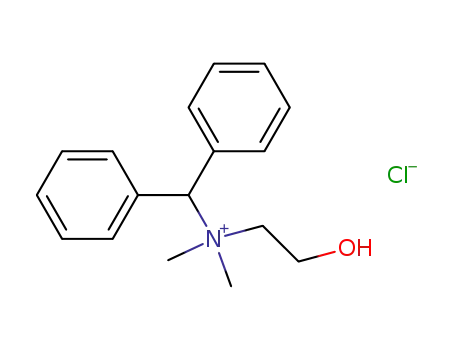
benzhydryl-(2-hydroxy-ethyl)-dimethyl-ammonium; chloride
-
90-99-3

diphenylchloromethane
-
108-01-0

2-(N,N-dimethylamino)ethanol
-
58-73-1

2-diphenylmethoxy-N,N-dimethylethanamine
147-24-0 Downstream products
-
108-01-0

2-(N,N-dimethylamino)ethanol
-
91-01-0
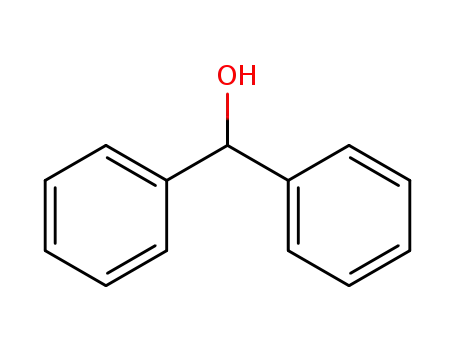
1,1-Diphenylmethanol
-
90-99-3

diphenylchloromethane
-
58-73-1

2-diphenylmethoxy-N,N-dimethylethanamine