CasNo:616-47-7
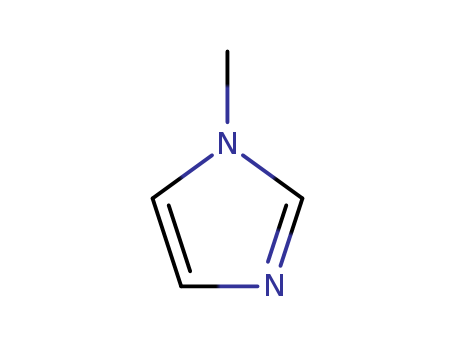
Product Name:1-Methylimidazole
Molecular Formula:C4H6N2
Appearance:colorless transparent liquid
Purity:99%
Factory Supply Sale 1-Methylimidazole 616-47-7 with Competitive Price
- Molecular Formula:C4H6N2
- Molecular Weight:82.105
- Appearance/Colour:colorless transparent liquid
- Vapor Pressure:0.4 mm Hg ( 20 °C)
- Melting Point:-60 °C(lit.)
- Refractive Index:n20/D 1.495(lit.)
- Boiling Point:198.679 °C at 760 mmHg
- PKA:6.95(at 25℃)
- Flash Point:92.222 °C
- PSA:17.82000
- Density:0.996 g/cm3
- LogP:0.42010
1-Methylimidazole(Cas 616-47-7) Usage
|
Uses |
1-Methylimidazole is an important raw material in the synthesis of pharmaceutical intermediates. It is used in the preparation of various pharmaceutical compounds, including losartan, nizofenone, 1-Methyl-1H-imidazole-5-carbonyl chloride hydrochloride, and naphazoline hydrochloride, among others. It is utilized as a specialty solvent in various chemical processes. 1-Methylimidazole can function as a base in chemical reactions and is often used to facilitate reactions involving acid-base chemistry. |
|
General Description |
1-Methylimidazole is typically a colorless to yellow liquid with an amine-like odor. It is miscible with water, which means it can be mixed with water in any proportion. Industrially, 1-Methylimidazole is prepared through two main routes. One method involves the acid-catalyzed methylation of imidazole using methanol. The second method is the Radziszewski reaction, which utilizes glyoxal, formaldehyde, ammonia, and methylamine as starting materials. |
|
Flammability and Explosibility |
Notclassified |
|
Purification Methods |
Dry it with sodium metal and then distil it. Store it at 0o under dry argon. The picrate has m 159.5-160.5o (from H2O). [Beilstein 23 III/IV 568.] |
InChI:InChI=1/C4H6N2/c1-6-3-2-5-4-6/h2-4H,1H3
616-47-7 Relevant articles
Ternary polymer electrolytes with 1-methylimidazole based ionic liquids and aprotic solvents
Jakub Reiter a 1, Jiří Vondrák a, Jiří Michálek b c, Zdeněk Mička d
, Electrochimica Acta Volume 52, Issue 3, 12 November 2006, Pages 1398-1408
New, defined way of preparation together with excellent chemical and electrochemical long-term stability of used compounds offers combination of acrylate-based polymer electrolytes with ionic liquids based on 1-methylimidazole.
Nitrosylmetalloporphyrins. III. Synthesis and molecular stereochemistry of nitrosyl-.alpha.,.beta.,.gamma.,.delta.-tetraphenylporphinato(1-methylimidazole)iron(II)
W. Robert Scheidt and Paul L. Piciulo
, J. Am. Chem. Soc. 1976, 98, 7, 1913–1919
Electronic effects resulting from coordination of the axial 1methylimidazole ligand could favor particular orientations of the nitric oxide; this could possibly reduce the range of …
Crystallographic and spectroscopic analysis of 9,10-bis-alkyl imidazolium anthracene hexatungstate supramolecular complexes
Annapareddy, Gayatri,Mubeena, Shaik,N, Meghana,Sarma, Monima
, (2021/06/28)
This article describes the ionic and sup...
616-47-7 Process route
-
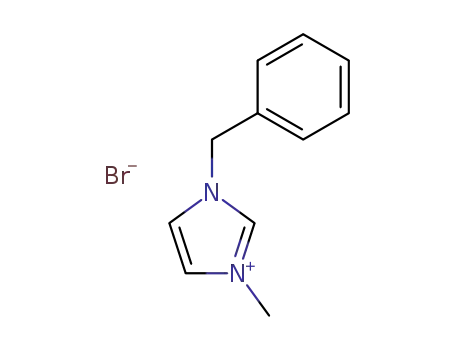
- 65039-11-4
3-benzyl-1-methylimidazolium bromide

-
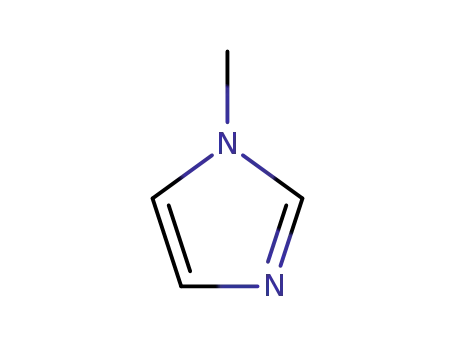
- 616-47-7
1-methyl-1H-imidazole

-
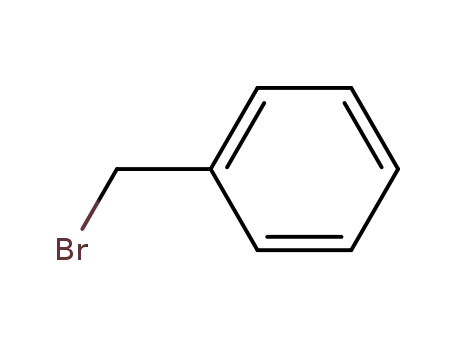
- 100-39-0
benzyl bromide
| Conditions | Yield |
|---|---|
|
at 260 - 300 ℃; under 12 Torr;
|
-
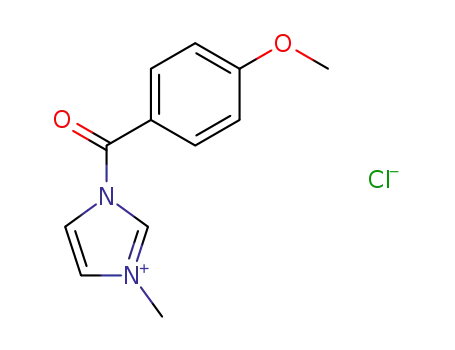
- 61166-15-2
1-(p-methoxybenzoyl)-3-methylimidazolium chloride

-

- 616-47-7
1-methyl-1H-imidazole

-
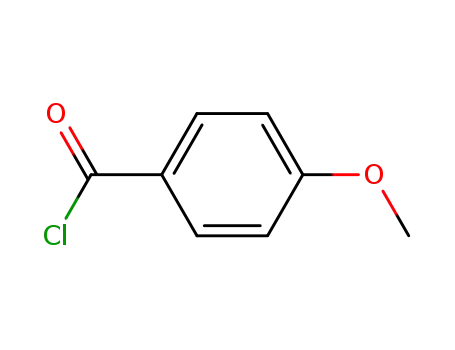
- 100-07-2
4-methoxy-benzoyl chloride
| Conditions | Yield |
|---|---|
|
In dichloromethane; at 25 ℃; Rate constant; Equilibrium constant;
|
616-47-7 Upstream products
-
288-32-4
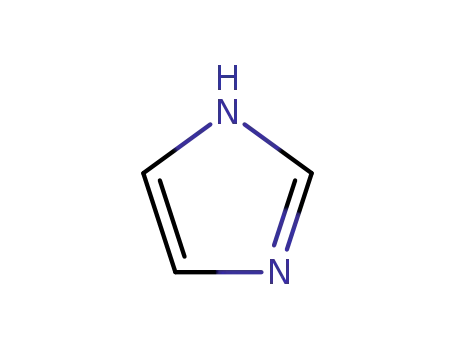
1H-imidazole
-
74-88-4
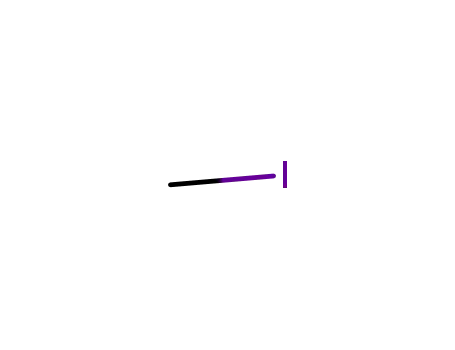
methyl iodide
-
872-49-1
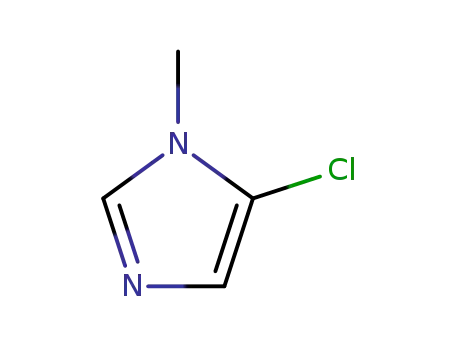
N-methyl-5-chloroimidazole
-
30148-21-1
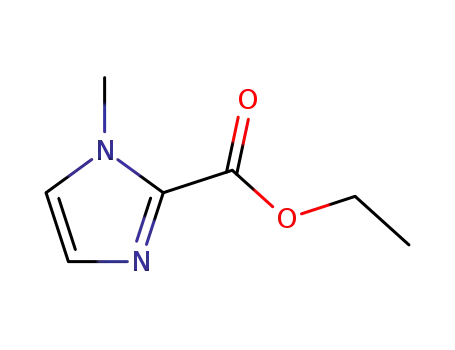
ethyl 1-methyl-1H-imidazole-2-carboxylate
616-47-7 Downstream products
-
41806-40-0
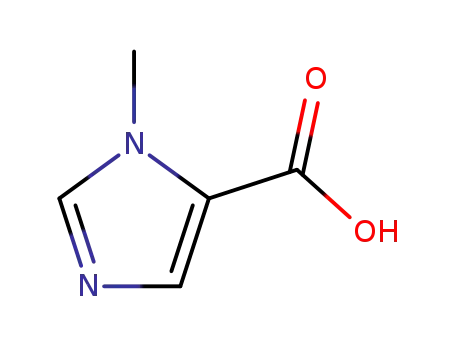
1-methyl-1H-imidazole-5-carboxylic acid
-
20485-43-2
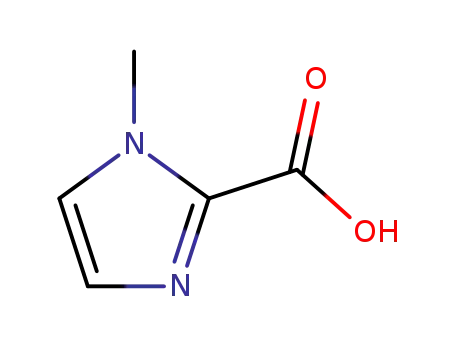
1-methyl-2-imidazolecarboxylic acid
-
17334-08-6
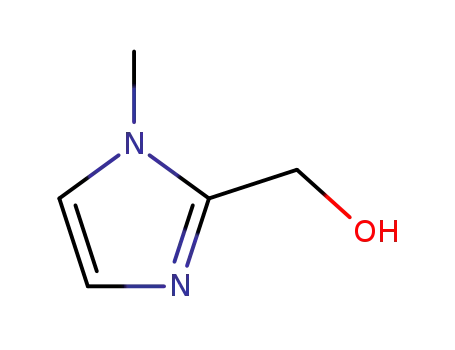
1-methyl-2-hydroxymethylimidazole
-
79917-89-8
1-methyl-3-propyl-1H-imidazolium chloride