CasNo:53716-49-7
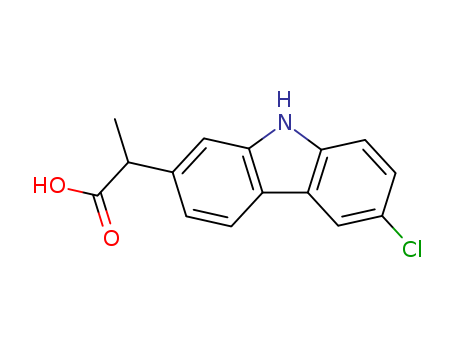
Product Name:Carprofen
Molecular Formula:C15H12ClNO2
Appearance:Off-white crystalline solid
Purity:99%
Chinese Manufacturer Supply Carprofen, Buy 53716-49-7 with Lowest Price
- Molecular Formula:C15H12ClNO2
- Molecular Weight:273.719
- Appearance/Colour:Off-white crystalline solid
- Vapor Pressure:3.45E-11mmHg at 25°C
- Melting Point:186-188 °C
- Refractive Index:1.731
- Boiling Point:509.1 °C at 760 mmHg
- PKA:4.84±0.30(Predicted)
- Flash Point:261.7 °C
- PSA:53.09000
- Density:1.42 g/cm3
- LogP:4.16260
Carprofen(Cas 53716-49-7) Usage
|
Description |
Carprofen is a nonsteroidal anti-inflammatory drug (NSAID) belonging to the carbazole and propionic acid class. It was initially developed for use in both humans and animals, but its use in humans has been discontinued. Today, it is primarily available for veterinary use and is prescribed by veterinarians to manage various conditions in animals, particularly dogs. |
|
Chemical Properties |
Off-White Crystalline Solid |
|
Originator |
Imadyl,Roche,Switz.,1981 |
|
Uses |
In veterinary medicine, Carprofen is prescribed to alleviate pain and inflammation caused by conditions such as osteoarthritis. It is also used to manage pain following surgical procedures in dogs. Carprofen is administered orally in the form of caplets.In Europe, it is reportedly registered for single-dose use in cats, but concerns have been raised about adverse effects like vomiting when used beyond a single dose. |
|
Therapeutic Function |
Antiinflammatory |
InChI:InChI=1/C15H12ClNO2/c1-8(15(18)19)9-2-4-11-12-7-10(16)3-5-13(12)17-14(11)6-9/h2-8,17H,1H3,(H,18,19)
53716-49-7 Relevant articles
Electrochemical Synthesis of Carbazoles by Dehydrogenative Coupling Reaction
Kehl, Anton,Schupp, Niclas,Breising, Valentina M.,Schollmeyer, Dieter,Waldvogel, Siegfried R.
, p. 15847 - 15851 (2020/11/02)
A constant current protocol, employing u...
Use of carprofen for the treatment of pain and inflammation in dogs.
Fox SM 1 , Johnston SA
Journal of the American Veterinary Medical Association, 01 May 1997, 210(10):1493-1498
Carprofen is a propionic acid-derived NSAID that has anti-inflammatory, analgesic, and antipyretic activity. In animals, carprofen is as potent as indomethacina and more potent than aspirin or phenlbutazone, but carprofen appears to be safer than most other NSAID.
Randomized, controlled trial of the efficacy of carprofen, a nonsteroidal anti-inflammatory drug, in the treatment of osteoarthritis in dogs.
Vasseur PB 1 , Johnson AL , Budsberg SC , Lincoln JD , Toombs JP , Whitehair JG , Lentz EL
, Journal of the American Veterinary Medical Association, 01 Mar 1995, 206(6):807-811
For force plate evaluation, the odds ratio was 3.3, meaning that a dog treated with carprofen was 3.3 times more likely to have a positive response than was a dog treated with the placebo. For evaluation by a veterinarian, the odds ratio was 3.5, and for owner evaluation, the odds ratio was 4.2. Institution where dogs were treated did not have a significant effect on results.
53716-49-7 Process route
-
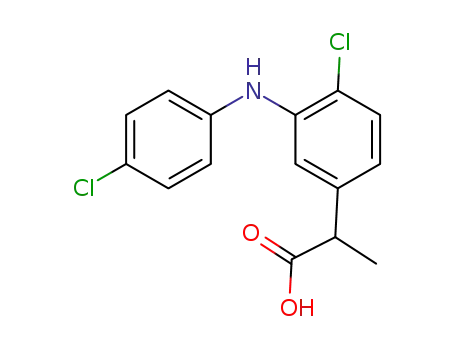
-
2-(3-((4-chlorophenyl)amino)-4-chlorophenyl)propanoic acid

-
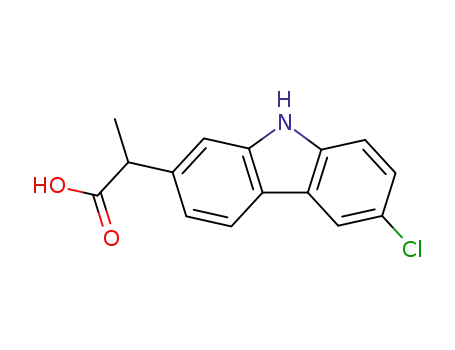
- 53716-49-7,52263-83-9
2-(6-chloro-carbazol-2-yl)-propionic acid
| Conditions | Yield |
|---|---|
|
With triphenylphosphine; palladium dichloride; sodium t-butanolate; In N,N-dimethyl-formamide; at 100 ℃; for 10h; Reagent/catalyst; Inert atmosphere;
|
91% |
|
2-(3-((4-chlorophenyl)amino)-4-chlorophenyl)propanoic acid; With triphenylphosphine; nickel dichloride; palladium dichloride; sodium t-butanolate; In N,N-dimethyl-formamide; at 100 ℃; for 10h; Inert atmosphere;
With acetic acid; In N,N-dimethyl-formamide; at 20 ℃; for 0.166667h;
|
87.9% |
|
With triphenylphosphine; nickel dichloride; palladium dichloride; sodium t-butanolate; In N,N-dimethyl-formamide; at 100 ℃; for 10h; Reagent/catalyst; Inert atmosphere;
|
87.9% |
|
With triphenylphosphine; nickel dichloride; palladium dichloride; sodium t-butanolate; In N,N-dimethyl-formamide; at 100 ℃; for 10h; Inert atmosphere;
|
87.9% |
-
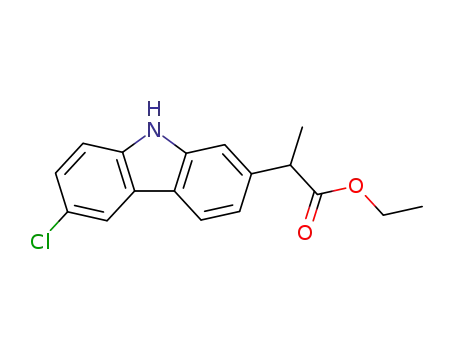
- 52262-89-2
ethyl 2-(6-chloro-9H-carbazol-2-yl)propanoate

-

- 53716-49-7,52263-83-9
2-(6-chloro-carbazol-2-yl)-propionic acid
| Conditions | Yield |
|---|---|
|
With water; sodium hydroxide; In methanol; at 20 ℃; for 8h; Inert atmosphere; Schlenk technique;
|
91% |
|
With sodium hydroxide; In methanol; water;
|
52% |