CasNo:2886-65-9
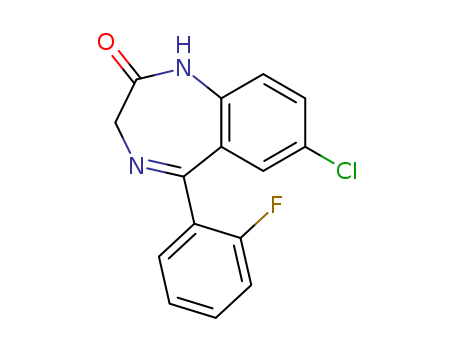
Product Name:N-desalkylflurazepam
Molecular Formula:C15H10ClFN2O
Appearance:Light yellow solid
Purity:99%
99% Pure Factory Supply High Purity N-desalkylflurazepam 2886-65-9 Safe Shipping
- Molecular Formula:C15H10ClFN2O
- Molecular Weight:288.709
- Appearance/Colour:Light yellow solid
- Vapor Pressure:1.97E-08mmHg at 25°C
- Melting Point:204-206 °C
- Refractive Index:1.647
- Boiling Point:454 °C at 760 mmHg
- PKA:11.55±0.70(Predicted)
- Flash Point:228.4 °C
- PSA:41.46000
- Density:1.39 g/cm3
- LogP:2.84220
7-Chloro-5-(2-fluoro-phenyl)-1,3-dihydro-2H-1,4-benzodiazepin-2-one(Cas 2886-65-9) Usage
|
Description |
Desalkylflurazepam (Item No. 18484) is an analytical reference material that is structurally categorized as a benzodiazepine. It is an active metabolite of several benzodiazepines, including flurazepam , flutoprazepam, fludiazepam, midazolam, and quazepam. Desalkylflurazepam inhibits L-type voltage-gated calcium channels (Cav; IC50s = 55 and 37 μM for Cav1.2 and 1.3, respectively) by positively modulating GABAA receptors. It can be detected in urine, serum, and meconium by LC-MS/MS. This product is intended for research and forensic applications. |
|
Chemical Properties |
Light Yellow Solid |
|
Uses |
The major human metabolite of flurazepam. This is a controlled drug precursor therefore a liscence may be required for purchase |
|
Mode of action |
N-Desalkylflurazepam is a benzodiazepine analog and an active metabolite of several other benzodiazepine drugs including flurazepam, flutoprazepam, fludiazepam, midazolam, flutazolam, quazepam, and ethyl loflazepate. It is long-acting, prone to accumulation, and binds unselectively to the various benzodiazepine receptor subtypes. It has been sold as a designer drug from 2016 onward. |
InChI:InChI=1/C15H10ClFN2O/c16-9-5-6-13-11(7-9)15(18-8-14(20)19-13)10-3-1-2-4-12(10)17/h1-7H,8H2,(H,19,20)
2886-65-9 Relevant articles
Improved and scalable methods for the synthesis of midazolam drug and its analogues using isocyanide reagents
Taghizadeh, Mohammad Javad,malakpouri, Gholam reza,Javidan, Abdollah
, p. 785 - 794 (2019/03/27)
Abstract: In this research, two improved...
One-pot microwave-assisted synthesis and antimalarial activity of ferrocenyl benzodiazepines
Mwande-Maguene, Gabin,Jakhlal, Jouda,Lekana-Douki, Jean-Bernard,Mouray, Elisabeth,Bousquet, Till,Pellegrini, Sylvain,Grellier, Philippe,Ndouo, Fousseyni Samba Toure,Lebibi, Jacques,Pelinski, Lydie
, p. 2412 - 2415 (2012/01/04)
An efficient synthesis of 1,4-benzodiaze...
Synthesis and in vitro anti-hepatitis B virus activities of 4-aryl-6-chloro-quinolin-2-one and 5-aryl-7-chloro-1,4-benzodiazepine derivatives
Cheng, Pi,Zhang, Quan,Ma, Yun-Bao,Jiang, Zhi-Yong,Zhang, Xue-Mei,Zhang, Feng-Xue,Chen, Ji-Jun
supporting information; scheme or table, p. 3787 - 3789 (2009/04/06)
A series of 4-aryl-6-chloro-quinolin-2-o...
Synthesis of?some?new substituted triazolo [4,3-a][1,4] benzodiazepine derivatives as?potent anticonvulsants
Narayana,Vijaya Raj,Ashalatha,Kumari, N. Suchetha
, p. 417 - 422 (2007/10/03)
Novel 8-chloro-6-(2-fluorophenyl)-1-(ary...
2886-65-9 Process route
-
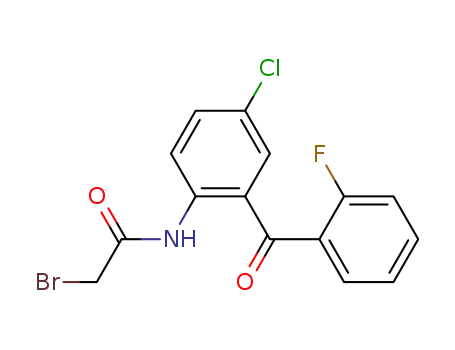
-
1584-62-9
2?(2?bromoacetamido)?5?chloro?2'?fluorobenzophenone

-
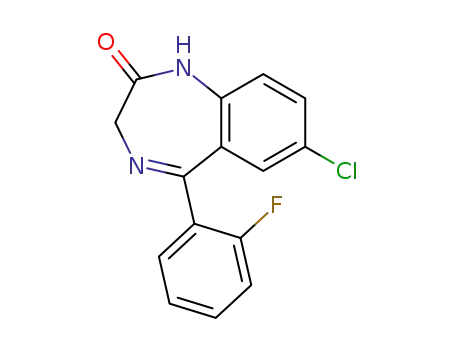
-
2886-65-9
7-chloro-5-(2-fluorophenyl)-1,3-dihydro-1,4-benzodiazepin-2(2H)-one
| Conditions | Yield |
|---|---|
|
With
ammonia;
In
methanol;
at 0 ℃;
Reflux;
|
70% |
|
With
ammonium hydroxide;
In
methanol; diethyl ether;
at 20 ℃;
for 46h;
|
|
|
With
ammonia;
In
methanol;
at 45 ℃;
|
18.65 g |
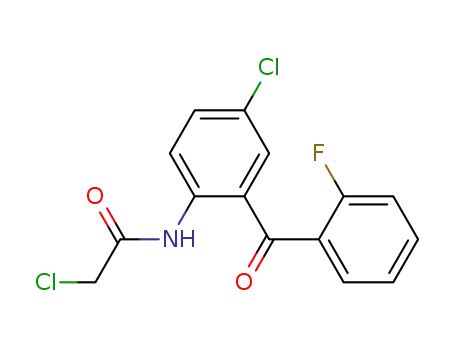
-
-
2836-40-0
2-(2-chloroacetyl)amino-4-chloro-2'-fluorobenzophenone

-

-
2886-65-9
7-chloro-5-(2-fluorophenyl)-1,3-dihydro-1,4-benzodiazepin-2(2H)-one
| Conditions | Yield |
|---|---|
|
With
paraformaldehyde;
In
methanol; ammonia; water;
|
71% |
|
With
hexamethylenetetramine; ammonium chloride;
In
ethanol;
for 4h;
Heating;
|
|
|
With
ammonium acetate; hexamethylenetetramine;
In
ethanol;
Reflux;
|
2886-65-9 Upstream products
-
76488-08-9
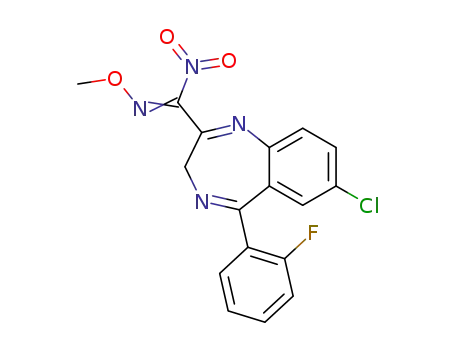
7-chloro-5-(2-fluorophenyl)-N-methoxy-alpha-nitro-3H-1,4-benzodiazepine-2-methanimine
-
1172-18-5
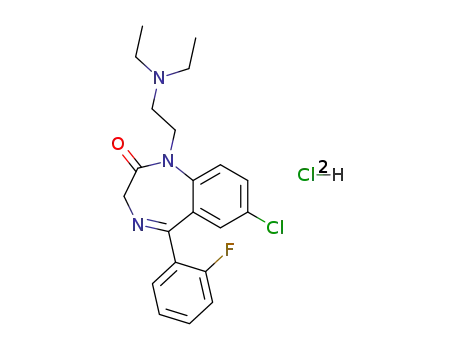
flurazepam dihydrochloride
-
17617-23-1
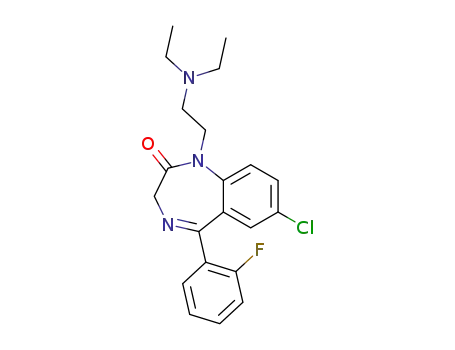
flurazepam
-
100-97-0
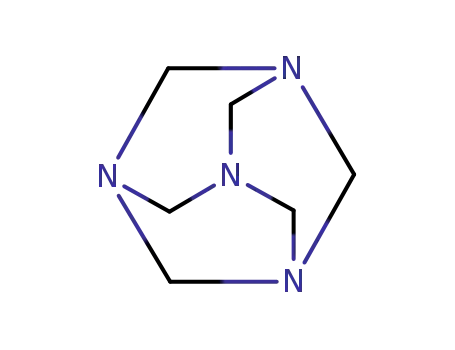
hexamethylenetetramine
2886-65-9 Downstream products
-
67974-53-2
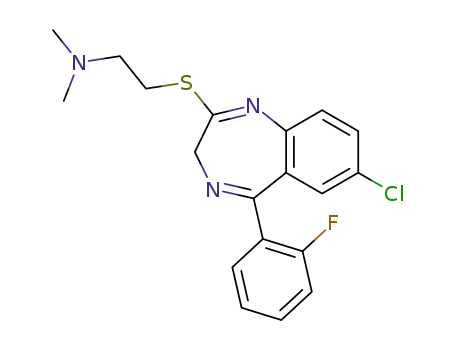
2-(2-dimethylaminoethylthio)-5-(2-fluorophenyl)-7-chloro-3H-1,4-benzodiazepine
-
112634-59-0
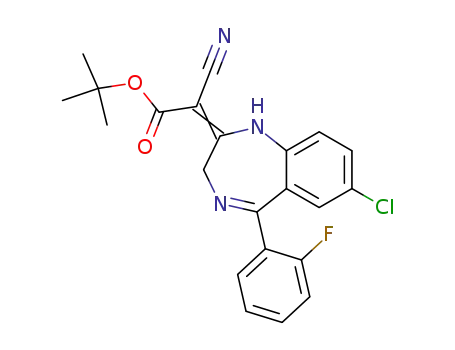
7-chloro-5-(2-fluorophenyl)-2,3-dihydro-1H-1,4-benzodiazepin-2-ylidenecyanoacetic acid t-butyl ester
-
1645-32-5
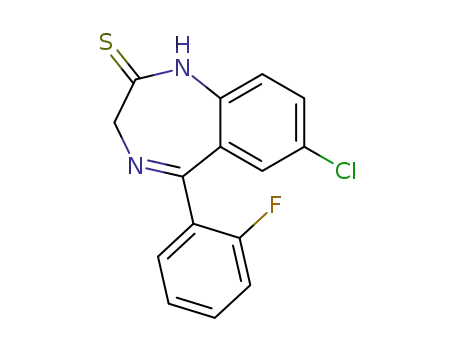
7-chloro-1,3-dihydro-5-(2-fluorophenyl)-2H-1,4-benzodiazepine-2-thione
-
72012-91-0
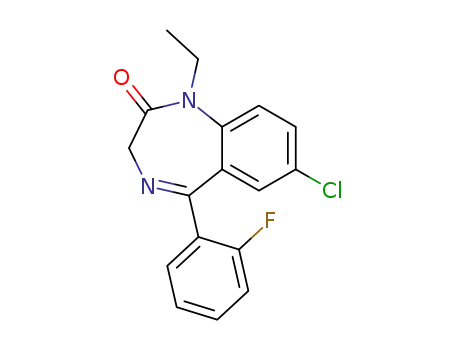
7-chloro-1-ethyl-(o-fluorophenyl)-3H-[1,4]benzodiazepin-2-one